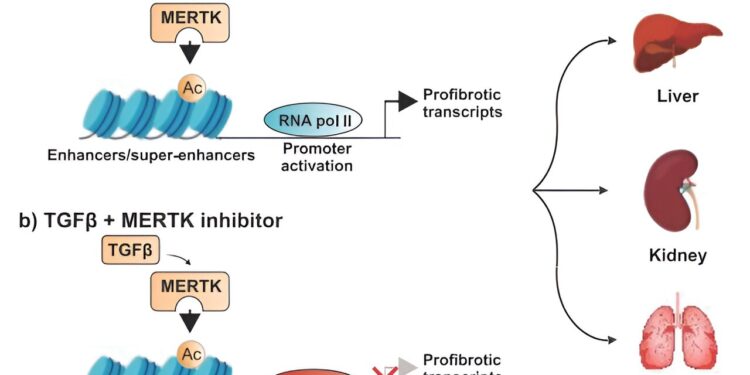
Model of MERTK mechanisms for regulation of the TGFβ core fibrotic pathway in various organs. Credit: Scientific translational medicine (2024). DOI: 10.1126/scitranslmed.adj0133
Medical science has long sought a deeper understanding of the devastating scarring of the body’s organs, known as fibrosis, which leads to irrevocable loss of function.
Fibrosis refers to the abnormal accumulation of connective tissue in organs, impairing their ability to function properly. This process is triggered by chronic inflammatory activity. These inflammatory processes can be induced by persistent infection, chemical insults, or severe tissue damage. In some patients, the underlying cause of their fibrotic disease remains stubbornly elusive. Fibrotic scars not only rob patients of healthy organ function, but in some cases, they rob them of their lives.
Scar tissue can damage any of the body’s major organs, manifesting in the lungs as idiopathic pulmonary fibrosis or in the liver as hepatic steatosis. Renal and cardiac fibrosis are conditions that can be characterized by severe organ damage. That is, a heart attack can cause a buildup of scar tissue (myocardial fibrosis), but the fibrotic area lacks the contractile activity of healthy heart tissue. Worse still, there are few therapies that can treat or permanently reverse organ fibrosis, making the search for potential therapies a top research priority.
Now, Australian medical researchers are uncovering new clues about fibrosis and how this damaging disease is sustained by a positive feedback loop in organs marked by pervasive fibrous scarring.
By isolating and defining the molecular activity of the feedback loop, Dr Ziyan Pan and colleagues at the Westmead Institute for Medical Research near Sydney have opened a new window of understanding into fibrosis by identifying a promising target for drug therapy.
In a study published in Scientific translational medicinePan and a team of collaborators emphasized that isolating the positive feedback loop was critical because it helped explain the persistent and sometimes deadly role that fibrosis can play in organ damage.
Tissue fibrosis is at the root of many chronic diseases that are difficult to treat, Pan and colleagues say. And while the cytokine known as transforming growth factor-β (TGFβ) regulates fibrosis, it is also involved in other critical biological processes, making it an unlikely target for drug development.
“TGFβ drives fibrosis and disease progression in a number of chronic disorders, but targeting this ubiquitously expressed cytokine may not result in a viable and safe antifibrotic therapy,” Pan writes in the journal.
“We sought to identify alternative ways to inhibit TGFβ signaling using human hepatic stellate cells and macrophages from humans and mice in vitro, as well as mouse models of liver, kidney, and lung fibrosis,” Pan continued.
Because TGFβ is a problematic potential target for drugs, Pan and his colleagues looked for a different, more durable target. In their study of mouse models of liver, kidney, and lung fibrosis, the researchers found that these organs had very high levels of an enzyme known as MERTK.
They also found that liver biopsy samples from patients with fibrotic fatty liver disease also contained high levels of MERTK, particularly samples from patients with significantly advanced stages of fibrosis.
What the team did next laid the foundation for a potential treatment strategy for fibrosis, regardless of which organ the disease attacks.
Returning to mouse models of liver, kidney, and lung fibrosis, Pan and the other scientists found that MERTK also induced TGFβ expression and stimulated TGFβ signaling, which set up the positive feedback loop that promoted fibrosis in cultured cells. In their study, the researchers found a way to attenuate MERT, stopping TGFβ signaling and the ongoing process of fibrosis.
“MERTK increased transcription of fibrosis-regulating genes by modulating chromatin accessibility and RNA polymerase II activity,” Pan wrote. “In all three mouse models, disrupting the fibrosis-promoting signaling loop by reducing MERTK expression reduced organ fibrosis.”
Scientists administered an experimental drug to the fibrotic cells, a MERTK inhibitor called UNC569. The experimental drug reduced fibrosis in the liver, kidneys, and lungs of mouse models when given early in the disease process. To Pan and the team’s surprise, the experimental drug also reversed liver fibrosis when given to mice with established liver damage.
“Pharmacological inhibition of MERTK reduced fibrosis in these mouse models, either when initiated immediately after injury or when initiated after fibrosis was established,” Pan said. “Together, these data suggest that MERTK plays a role in modulating organ fibrosis and may be a potential target for the treatment of fibrotic diseases.”
Whether the experimental MERTK inhibitor known as UNC569 will become a drug for human testing remains to be seen. But more importantly, Pan and his colleagues have shown that fibrosis is maintained in tissues by the same positive feedback loop, and that this loop can be disrupted by drugs.
“These results expand our understanding of the roles of MERTK, but also reveal novel regulatory molecules that regulate TGF-β activity during fibrosis,” Pan concluded.
More information:
Ziyan Pan et al, MERTK inhibition reduces organ fibrosis in mouse models of fibrotic disease, Scientific translational medicine (2024). DOI: 10.1126/scitranslmed.adj0133
© 2024 Science X Network
Quote:Fibrosis Lacks Treatments and Reversal Methods: Study Identifies Promising Target for Drug Therapy (2024, September 23) Retrieved September 23, 2024 from
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. The content is provided for informational purposes only.