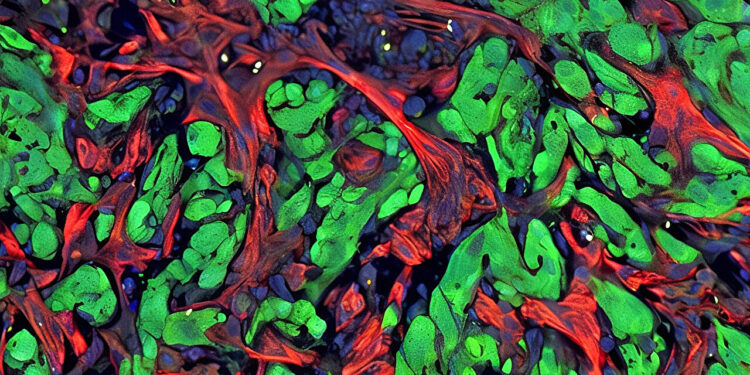
Tissue section from a mouse model of lung metastasis showing cancer cells (green) that have attracted and activated fibroblasts (red) to support the growth of a metastatic tumor. Credit: Massagué Lab/ Memorial Sloan Kettering Cancer Center
When it comes to cancer metastasis, it takes two to tango. That’s one of the key findings of a new study led by researchers at Memorial Sloan Kettering Cancer Center (MSK): TGF-beta and RAS signaling pathways work together to drive cancer spread in lung adenocarcinoma, a leading cause of cancer death worldwide.
Remove one of these two signals and lung cancer will not be able to spread (metastasize) to new parts of the body, their findings in animal models suggest.
The study, published on September 6 in Cellindicates new opportunities to potentially prevent metastases, thanks to an updated understanding of the underlying processes.
“About 9 out of 10 cancer deaths are caused by metastasis,” said Jun Ho Lee, Ph.D., first author of the study and a postdoctoral researcher in the lab of the study’s senior author, Joan Massagué, Ph.D. “So research aimed at understanding, preventing, and treating metastasis has great potential to improve the lives of many people.”
A tale of two signaling pathways: TGF-Beta and RAS
By itself, TGF-beta, a type of signaling protein known as a cytokine, is not an ideal drug target. That’s because it plays so many important roles in the body, making it nearly impossible to block without risking major side effects.
TGF-beta normally plays a key role in programs that regulate embryonic development and injury repair. But these programs are also activated in cancer cells during metastasis, giving otherwise immobile cells the ability to move and invade tissues.
However, in this case, metastases also require input from the RAS pathway, the researchers found.
The normal role of RAS is to transmit signals from outside a cell to the cell nucleus, where it activates genes that control cell growth, division, and differentiation.
Researchers have determined that a transcription factor (a protein that specifically regulates the activity of certain genes) controlled by RAS plays a critical role in metastasis. It is known as RAS response element binding protein 1 (RREB1).
They found that RREB1 collaborates with a signaling complex called SMAD4, which is controlled by TGF-beta. Inhibiting RREB1 disabled the metastatic process in mouse models, suggesting that it could be a potential new drug target, both in lung cancer and in related diseases such as pulmonary fibrosis.
Why context is key for the role of TGF-Beta in cancer metastasis
The study brings together two long-standing research areas of the Massagué lab: TGF-beta signaling and metastasis.
Dr. Massagué, who is Director of the Sloan Kettering Institute and Chief Scientific Officer of MSK, is known for his pioneering work in uncovering the mechanism of action of transforming growth factor beta (TGF-beta) signaling.
The TGF-beta family of proteins plays a key role in the proliferation and differentiation of many different cell types, controlling the formation and regeneration of tissues from embryonic life to adulthood.
“We know that the TGF-beta pathway promotes injury repair and suppresses tumor formation in healthy tissue, but when tumors manage to grow, it promotes metastasis,” explains Dr. Massagué.
“So that’s a big question that we wanted to understand: How is it that metastases can take something that’s normally there to keep tissues healthy, transform it, and use it to promote metastatic growth? And that’s led to a whole research program in my lab.”
Another way to put it is that TGF-beta helps normal cells regenerate, but when it is taken over by cancer, that regeneration turns into uncontrollable growth instead of healthy renewal.
In this study, the team shed new light on the mechanisms at work in this process, revealing that the contributions of RAS and TGF-beta are necessary for the take-off of metastases, at least in this most common form of lung cancer, where the growth of fibrous scar tissue is a key element.
Beyond cancer, TGF-beta-induced pulmonary fibrosis affects hundreds of thousands of people worldwide. While targeting TGF-beta is considered promising, long-term use of TGF-beta inhibitors to treat metastatic cancer or fibrosis could lead to significant side effects. For example, disrupting TGF-beta’s work can lead to excessive immune system activity, Dr. Massagué notes.
“So the biotech industry has been looking for ways to target TGF-beta in a partial or restricted way,” he says. “Here we have discovered a new candidate for this type of intervention, focused only on a partner signal that we now know is also required for metastasis and fibrosis.”
Better understanding the ability of cancer to spread
The study is part of broader efforts in the MSK field to shed new light on how cancer progresses and spreads.
“By studying the interactions between cancer and the environment in which it develops, what we call the ‘tumor microenvironment,’ we are beginning to discover how cancer programs are determined by specific signals, involving pre-established response patterns that modify the behavior of the cancer cell and neighboring cells,” explains Dr. Massagué.
In this case, neighboring cells include fibroblasts that produce collagen fibers.
Additionally, studies like this one, which seek to understand the forces behind metastasis, further reveal how overall tissue maintenance and regeneration after injury is tightly organized and orchestrated, Dr. Massagué says.
“We can then extend this knowledge to other types of tumors, to other organs in the body where metastases develop, to other forms of fibrosis,” he says.
More information:
Jun Ho Lee et al, TGF-β and RAS jointly unmask activators primed to promote metastasis, Cell (2024). DOI: 10.1016/j.cell.2024.08.014. www.cell.com/cell/fulltext/S0092-8674(24)00905-X
Cell
Provided by Memorial Sloan Kettering Cancer Center
Quote:Study finds that both TGF-beta and RAS signaling are required for lung cancer metastasis (2024, September 6) retrieved September 6, 2024 from
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. The content is provided for informational purposes only.