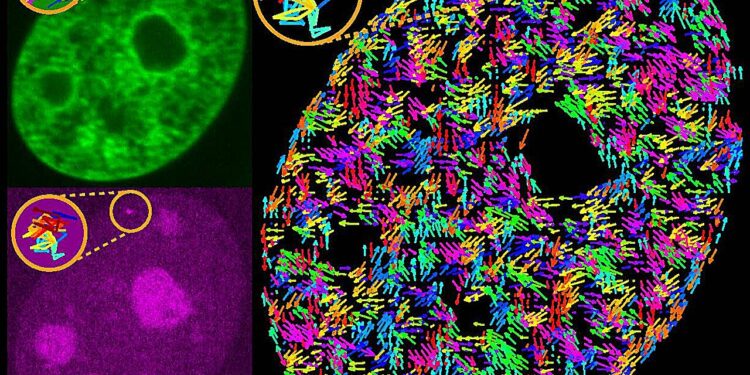
Visualization of the genome (green) and a single gene (magenta) in the nucleus of a human cell, with genomic movements mapped by arrows and an example of a genetic trajectory highlighted as a colored curve. Credit: Alexandra Zidovska, Department of Physics, NYU.
A team of scientists has discovered surprising links between gene activity, genome packaging and genome-wide movements, revealing aspects of genome organization that directly affect the regulation and expression of Genoa.
The results, reported in the journal Natural communicationsadvance understanding of the mechanisms behind transcription-dependent single gene movements, dysfunction of which can lead to neurological and cardiovascular disorders as well as cancer.
“The genome is ‘jiggled’ by transcription-driven movements of single genes,” says Alexandra Zidovska, professor of physics at New York University and lead author of the study.
“Genes move differently depending on whether they are read or not, leading to complex and turbulent movements of the human genome. Understanding the mechanisms behind transcription-dependent movements of single genes in the nucleus could be key to understanding the human genome in health and disease.
The human genome is made up of two meters (six and a half feet) of DNA, which is packaged inside the cell in a nucleus just 10 micrometers in diameter, or 100,000 times smaller than the length of the DNA of the genome. The DNA molecule encodes information for all cellular processes and functions, with genes serving as the units of information.
Different genes are read and their information processed at different times. When a gene is read, molecular machinery accesses it and transcribes its information into an mRNA molecule, a process called transcription.
Zidovska and her colleagues previously discovered that the genome undergoes numerous “wiggles,” or movements, leading to its reorganization and repositioning in the nucleus.
However, the origin of these movements is little understood. Scientists have hypothesized that molecular motors powered by adenosine triphosphate (ATP) molecules, which provide energy for many biological processes, are the driving forces.
These active motors are thought to apply forces to the DNA, which can result in movement of the DNA and the nucleoplasm, its surrounding fluid. But the larger physical machinations behind this remain elusive.
With this in mind, Zidovska and her colleagues focused on RNA polymerase II, responsible for transcription and one of the most abundant molecular motors in the cell nucleus. When a gene is active, that is, actively transcribed, the molecular machinery responsible applies forces to the DNA during its processing.
THE Natural communications The study investigated how the movement of a single actively transcribed gene affects the movements of the genome surrounding it in living human cells.
To do this, the authors used CRISPR technology to fluorescently label single genes, high-resolution two-color microscopy of live cells to visualize the movement of these marked genes, and displacement correlation spectroscopy (DCS) to simultaneously map the flow of the genome through the nucleus. .
The high-resolution imaging data was then processed through physical and mathematical analysis, revealing a never-before-seen physical picture of how genes move inside the cell.
In their study, the researchers first looked at the movements of genes when they were inactive, then “turned on” these genes and observed how their movement changed once they were “active”. At the same time, the authors used DCS to map the flow of the surrounding genome, monitoring how the genome flows through the nucleus before and after gene activation.
Overall, the authors found that active genes contribute to the wiggle movement of the genome. Through simultaneous mapping of single gene and entire genome movements, they reveal that genome compaction affects gene contribution.
Specifically, movement correlation analysis indicated that a single active gene drives genome movement in low-compaction regions, but a high-compaction genome drives gene movement regardless of its activity state. .
“By revealing these unexpected links between gene activity, genome compaction and genome-wide movements, these results reveal aspects of the spatio-temporal organization of the genome that have a direct impact on the regulation and gene expression,” says Zidovska.
The work also adds to our understanding of physics.
“This research provides new knowledge about the physics of active and living systems,” she observes. “By revealing emergent behavior of active living systems, such as the human genome, it teaches us new physics.”
Other authors of the paper were Fang-Yi Chu and Alexis S. Clavijo, doctoral students at NYU, and Suho Lee, a postdoctoral researcher at NYU.
More information:
Transcription-dependent mobility of single genes and genome-wide movements in living human cells, Natural communications (2024). DOI: 10.1038/s41467-024-51149-4
Provided by New York University
Quote: Scientists discover how transcription drives movement within the genome (October 22, 2024) retrieved October 22, 2024 from
This document is subject to copyright. Except for fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for informational purposes only.