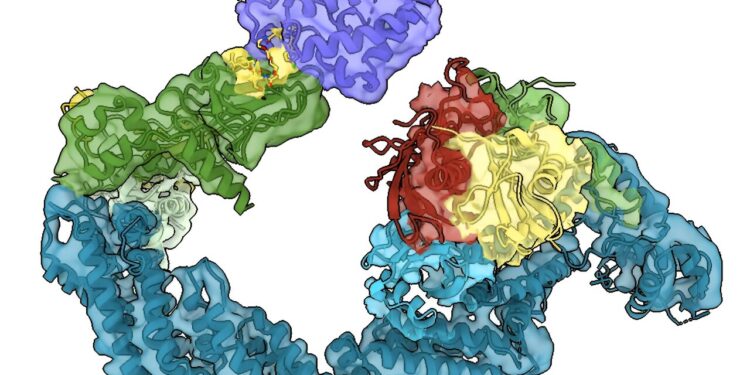
Visual of the MZ1 molecule. Credit: University of Dundee
Researchers at the University of Dundee have revealed in great detail the workings of molecules called protein degraders which can be deployed to combat what were previously considered “incurable” diseases, including cancers and neurodegenerative diseases.
Protein-degrading molecules herald a revolution in drug discovery, with more than 50 such drugs currently being tested in clinical trials for patients with diseases for which no other options exist.
The Center for Targeted Protein Degradation (CeTPD) at the University of Dundee is one of the world’s leading research centers into how protein degraders work and how they can be used most effectively for a new generation of medicines.
Researchers have now revealed previously invisible levels of detail and understanding of how protein degraders work, enabling even more targeted use of them at the molecular level.
PhD student Charlotte Crowe and Dr Mark Nakasone, senior postdoctoral researcher at CeTPD, used a technique called cryo-electron microscopy (cryo-EM), which allows scientists to see how biomolecules move and interact with each other.
It works by freezing proteins and using a focused electron beam and a high-resolution camera to generate millions of 2D images of the protein. They then used sophisticated software and artificial intelligence (AI) models that allowed them to generate 3D snapshots of the degrading drugs in action.
Their latest research is published in the journal Scientific advances and is expected to constitute a landmark contribution to research in the area of TPD and ubiquitin mechanisms.
“We have reached a level of detail that allows us to see how these protein degraders work and can be deployed (to recruit the pathogen protein) and target the target in molecular terms,” said Charlotte Crowe, who carried out the research in collaboration with a wider team of researchers in Dundee.
“Protein-degrading molecules work in a fundamentally different way than conventional drugs. However, until recently, the exact details of how this process works at the molecular level had remained elusive.
“Proteins generally measure a few nanometers, which corresponds to 1 billionth of a meter, or 1 millionth of the width of a hair. So it was not possible until now to be able to “see” them in action.
“We are now able to create an animated picture of how it all happens, meaning we can more specifically control the process in an incredible level of detail.”
Professor Alessio Ciulli, Director of CeTPD, said: “This is incredibly exciting work that opens up the possibility of developing even more effectively targeted drugs, capable of finally treating certain diseases that, until now, have been too difficult to fight. »
How it works
Proteins are essential for our cells to function properly, but when they don’t work properly, they can cause disease.
Targeted protein degradation involves repurposing protein recycling systems in our cells to destroy disease-causing proteins. Protein degraders work by capturing the disease-causing protein and making it stick like glue to the cellular protein recycling machinery, which then marks the protein as expired in order to destroy it.
The tag is a small protein called ubiquitin, which is effectively fired at the pathogen protein like a bullet. For the process to work effectively, the ubiquitin must reach the right places on the target protein so that it is tagged effectively. The Dundee team’s new work allows them to see how the ball hits the proverbial target.
Working with a protein-degrading molecule called MZ1, developed in the Ciulli laboratory in Dundee, and using high-end mass spectrometry, they were able to identify exactly where on the target protein the vital ‘tags’ are added.
The work shows how degrading drugs retain and position disease-causing proteins, making them good targets for receiving ubiquitin molecules (i.e. “ubiquitin-atables”), which then leads to their destruction inside the cell.
The efficiency and productivity of protein degradation depends on the ability of the degrading molecule to cling to the pathogenic protein and in which position it can act most effectively. This latest research draws a target and keeps it stable enough for the molecule to be targeted precisely.
Professor Ciulli said this and other recently published papers contribute to the rapid development of an exciting area of science and drug discovery. “This rapidly expanding field is fascinating, and complementary papers on how this cellular protein recycling machinery works to pull ubiquitin molecules onto target proteins were recently published from the laboratories of biochemists Brenda Schulman (Max Institute). Planck of Biochemistry) and Gary Kleiger (University of Nevada, Las Vegas).
“Our collective work represents a step forward in understanding that will accelerate the development of new TPD drugs in the future.”
This work comes from a local collaboration between two groups of scientists from the University of Dundee.
At the Center for Targeted Protein Degradation, led by Professor Alessio Ciulli, were Charlotte Crowe, Mark Nakasone, Conner Craigon, Gajanan Sathe and Nikolai Makukhin. They worked with Professor Ron Hay, an ubiquitin expert based at the School of Life Sciences, and his colleagues Sarah Chandler and Mike Tatham.
More information:
Charlotte Crowe et al, Mechanism of ubiquitinability of proteins targeted to degraders, Scientific advances (2024). DOI: 10.1126/sciadv.ado6492. www.science.org/doi/10.1126/sciadv.ado6492
Provided by the University of Dundee
Quote: Targeting ‘incurable’ diseases: Researchers reveal new levels of detail in targeted protein degradation (October 11, 2024) retrieved October 11, 2024 from
This document is subject to copyright. Except for fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for informational purposes only.