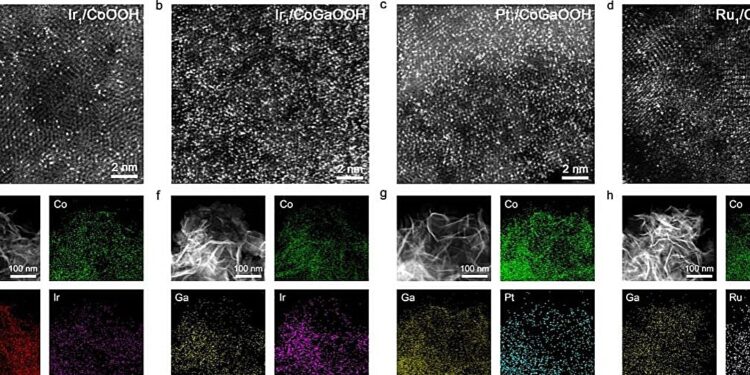
Structural images of single atoms at high density. Credit: Prof. BAO’s team
Hydrogen, with its green, low-carbon, and high-calorific properties, is emerging as the new solution to solve the energy crisis. Electrochemically, water splitting has attracted much attention as a promising technique for hydrogen production. However, the slow kinetics of the oxygen evolution reaction (OER) at the anode limits the overall energy conversion efficiency. Therefore, the development of efficient catalysts for OER is highly necessary.
Recently, a research team led by Professor Bao Jun from the University of Science and Technology of China (USTC) revealed that the high-density cobalt-based Ir single-atom catalyst supported by oxide exhibits excellent performance in electrochemical OER, which is closely related to the neighboring synergistic interactions in neighboring single atoms. The research was published in Applied Chemistry.
The performance of single-atom catalysts (SACs) is closely related to the density of single atoms on the surface. As the density of single atoms increases, the distance between atoms decreases, leading to the emergence of neighborhood synergy, which optimizes the adsorption behavior of reaction intermediates and subsequently improves the overall performance of the catalyst. Therefore, constructing high-density single atoms is an effective strategy to improve catalyst performance.
To achieve precise fabrication of high-density single atoms, the team introduced Ga atoms into the cobalt-based oxide lattice, thereby modulating the electronic structure of the single atom anchoring sites. This approach significantly enhanced the binding strength between the oxygen defect sites and the single atom precursors, successfully constructing a series of high-density SACs.
High-density Ir single atom neighborhood synergy mechanism in regulating REL performance. Credit: Professor Bao’s team
The team evaluated the OER performance of these catalysts to study the neighboring synergistic effects of high-density single atoms. The results showed that the high-density single atom Ir catalyst Nei-Ir1/CoGaOOH exhibited a low overpotential of 170 mV at a current density of 10 mA cm-2and long-term stability over 2000 h. In addition, the catalyst operated stably at a current density of 1 A cm-2 in an alkaline electrolyte for more than 50 hours. In situ Raman spectra confirmed that the catalyst maintained its structural stability during OER.
Further mechanism studies revealed that the improved catalyst performance does not come from the optimization of the electronic structure of the active sites, but rather from the synergistic neighborhood interaction of high-density Ir single atoms. The synergistic neighborhood interaction stabilizes the *OOH intermediates through additional hydrogen bonding interactions, thereby reducing the reaction energy barrier and significantly improving the catalyst performance.
This research unveils the mechanism underlying the improved performance of high-density monatomic Ir catalysts, providing new insights into the future development of electrochemical OER catalysts.
More information:
Peiyu Ma et al., Neighborhood synergy in high-density single IR atoms on CoGaOOH for efficient alkaline electrocatalytic oxygen evolution, Applied Chemistry (2024). DOI: 10.1002/ange.202404418
Provided by University of Science and Technology of China
Quote:Researchers reveal performance enhancement mechanism in single-atom catalyst for oxygen evolution reaction (2024, September 9) retrieved September 9, 2024 from
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. The content is provided for informational purposes only.