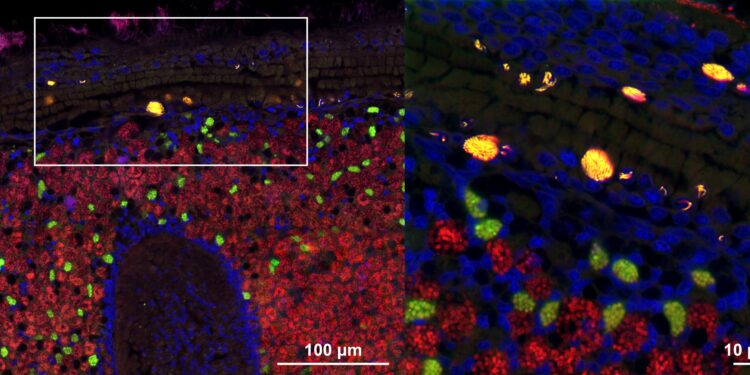
Microscopic image (confocal fluorescence in situ hybridization (FISH) microscopy) of tissues of a deep-sea mussel showing the intranuclear parasite Ca. Endonucleobacter in yellow and beneficial symbiotic bacteria in green and red. Cell nuclei are colored blue. The right panel is a zoom in of the white square in the left panel. Credit: Miguel Angel Gonzalez-Porras/Max Planck Institute for Marine Microbiology
Most animals live in close relationship with bacteria. Some of them live inside the cells of their hosts, but very few are able to live inside cell organelles (structures inside the cell, such as body organs). One group of bacteria has discovered how to colonize the nucleus of their hosts, a remarkable feat given that the nucleus is the control center of the cell.
Until now, nothing is known about the molecular and cellular processes used by these intranuclear bacteria to infect and reproduce in animal hosts. A group of scientists from the Max Planck Institute for Marine Microbiology in Bremen, Germany, now presents the first in-depth analysis of an intranuclear parasite of animals in a study published in Microbiology of nature.
How to reproduce massively in a cell without killing it
This intranuclear parasite, Candidatus endonucleobacter, infects the nuclei of deep-sea mussels from hydrothermal vents and cold springs worldwide. A single bacterial cell enters the mussel nucleus and then reproduces to over 80,000 cells, causing the nucleus to swell to 50 times its original size.
“We wanted to understand how the bacteria infect and reproduce inside the nuclei, and in particular how these bacteria acquire the nutrients they need for their massive replication, while preventing their host cells from dying,” explains Niko Leisch, co-lead author with Nicole Dubilier from the Symbiosis Department at the Max Planck Institute for Marine Microbiology.
Using a range of molecular and imaging methods, the scientists revealed that Ca. endonucleobacter feeds on sugars, lipids, and other cellular components of its host. It does not digest its host’s nucleic acids, as many other intranuclear bacteria do. This feeding strategy ensures that the host cell functions long enough to provide Ca. endonucleobacter with the nutrients it needs to reproduce in such large numbers.
Deep-sea Bathymodiolus mussels are found worldwide, in hydrothermal vents and cold springs. The mussels live in symbiosis with beneficial bacteria that provide them with food. The mussels also have a pathogenic bacterium that infects their nuclei. Interestingly, the only cells that are not infected by the pathogen are those that contain symbiotic bacteria. Credit: MARUM, University of Bremen
Arms Race for Cell Control
A common response of animal cells to infection is apoptosis, a suicide program that cells initiate when damaged or infected by bacteria or viruses.
“Interestingly, these bacteria have developed a sophisticated strategy to prevent their host cells from self-destructing,” says first author Miguel Ángel González Porras. “They produce proteins that suppress apoptosis, called inhibitors of apoptosis (IAPs).”
An arms race then ensues to control cell death: as the bacteria produce more and more IAP, the host cell ramps up its production of proteins that induce apoptosis. Eventually, after the parasite has had time to multiply en masse, the host cell ruptures, releasing the bacteria and allowing them to infect new host cells.
Dubilier adds: “The discovery of IAPs in Ca. endonucleobacter was one of the most surprising results of our study, because these proteins are only known in animals and a few viruses, but have never been found in bacteria.”
The authors’ analyses of the evolutionary relationships of IAPs revealed that the parasite likely acquired these genes from its host through horizontal gene transfer (HGT). While horizontal gene transfer from bacteria to eukaryotes is well known, only a few examples of horizontal gene transfer in the opposite direction, as the authors have now discovered, are known.
The implications of evolution for medicine
“Our discovery expands our understanding of host-microbe interactions and sheds light on the complex strategies that parasites have evolved to thrive in their hosts,” Dubilier says. These findings could have broader implications for the study of parasitic infections and immune evasion strategies in other organisms.
“Our research sheds light on a neglected mechanism of genetic exchange – the transfer of high-molecular-weight genes from eukaryotes to bacteria – that could influence our understanding of microbial evolution and pathogenesis. Furthermore, our study provides insights into the regulation of apoptosis, which is relevant for cancer research and cell biology,” Leisch concludes.
More information:
An intranuclear bacterial parasite of deep-sea mussels expresses host-acquired apoptosis inhibitors, Microbiology of nature (2024). DOI: 10.1038/s41564-024-01808-5
Provided by the Max Planck Society
Quote: Hijacking the Cell Command Center: Nuclear Parasites in Deep-Sea Mussels (2024, September 6) Retrieved September 6, 2024 from
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. The content is provided for informational purposes only.