Scientists have achieved the first total enantioselective synthesis of (–)-gukulenin A. Credit: Science (2025). DOI:10.1126/science.aea9310
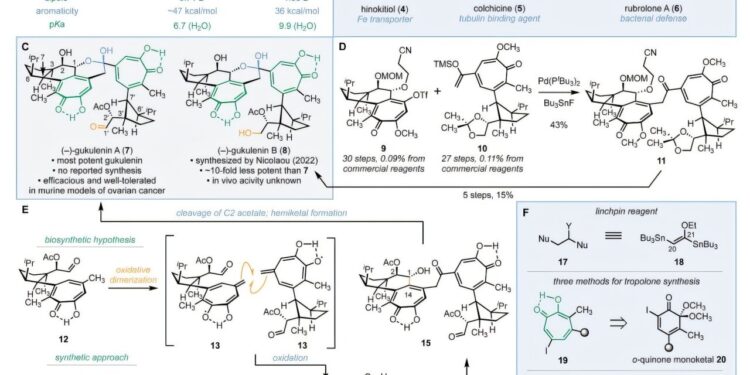
A team of researchers from Yale University, USA, successfully achieved the first stereoselective synthesis of the complex natural product (-)-gukulenin A (7), which exhibits notable cytotoxicity against ovarian cancer.
(–)-Gukulenin A is known to push chemists to their limits due to its challenging molecular motifs: two α-tropolones which are seven-membered aromatic rings with powerful molecular dipoles, 10 precise 3D stereocenters, and fragile chemical groups like a hemiketal and an aldehyde.
The researcher overcame the synthesis barrier with a clever three-component assembly strategy inspired by biosynthetic pathways. After successfully constructing the molecule, they designed 15 other derivatives to identify the structural features behind the cytotoxic potency of (–)-gukulenin A.
Their research led them to dimeric α-tropolones, at least 10 times more potent than their monomeric counterparts and, in some cases, 200 times more potent.
The results are published in Science.
Hidden in the sponge
Since the 1950s, when the unique molecular structures of α-tropolones were theorized, synthetic chemists have been captivated by these molecules.
It was first a matter of demystifying the structure of the molecule, then of studying its chemical properties and finding its derivatives in nature.
Over the years, researchers have extracted a wide range of α-tropolones from tree bark, flowers and bacteria. The (–)-gukulenin A family of molecules was discovered in the marine sponge Phorbas gukulensis, collected near Gageodo Island, South Korea.
Soon after, researchers discovered that gukulenins possessed remarkable biological activity. (–)-gukulenin A, in particular, emerged as a potent cytotoxin, reducing the size of ovarian tumors by more than 92% in mouse studies. Unlike many largely cytotoxic natural products, it demonstrated notable selectivity and was well tolerated in animal models.
The ability to synthesize this molecule on a large scale could open the door to new therapeutic options for ovarian cancer, but the delicate synthesis process has long hindered this goal.
The team created 15 derivatives to probe the structural features behind the cytotoxicity.Credit: Science (2025). DOI:10.1126/science.aea9310
The three-component assembly
In this research, the team adopted a three-step strategy to address the synthesis challenge: building the two halves, putting those halves together, and closing the final rings.
For the first part, they started with the readily available exo-2-norbornylamine, a rigid bicyclic molecule that naturally drove the 3D arrangement. They then developed a new ring expansion method to transform a 6-membered ring into the 7-membered tropolone structure needed for monomers. The two monomers were finally joined using a newly created and previously unknown two-carbon binding reagent: (E)-1,2-di(tributylstannyl)-1-ethoxyethylene.
The final step was to close the delicate hemimetal ring. Remarkably, the team achieved this simply by heating the intermediate to 120°C, triggering a precise reaction that closed the ring.
After successfully producing (–)-gukulenin A, the researchers used the same procedure to design 15 other derivatives. They tested and compared their cytotoxicity on four human cancer cell lines: lung, colon, leukemia and ovarian.
While the potency of gukulenin A varied across cancer types, the trend that remained consistent was that molecules with dimeric α-tropolone rings were at least 10 times more potent than monomeric molecules.
The researchers suggest that the cytotoxic potency may come from α-tropolone’s affinity for divalent metals, allowing (–)-gukulenin A to bind to two distinct metal-containing proteins simultaneously.
This information can help lay the foundation for determining the targets of (–)-gukulenin A (7) and set the stage for the preclinical evaluation of its synthetic derivatives as anticancer agents.
Written for you by our author Sanjukta Mondal, edited by Sadie Harley, and fact-checked and edited by Robert Egan, this article is the result of painstaking human work. We rely on readers like you to keep independent science journalism alive. If this reporting interests you, consider making a donation (especially monthly). You will get a without advertising account as a thank you.
More information:
Vaani Gupta et al, Three-component assembly and structure-function relationships of (–)-gukulenin A, Science (2025). DOI: 10.1126/science.aea9310
© 2025 Science X Network
Quote: New strategy for synthesizing (–)-gukulenin A reveals the chemistry behind its anticancer effects (November 18, 2025) retrieved November 18, 2025 from
This document is subject to copyright. Except for fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for informational purposes only.