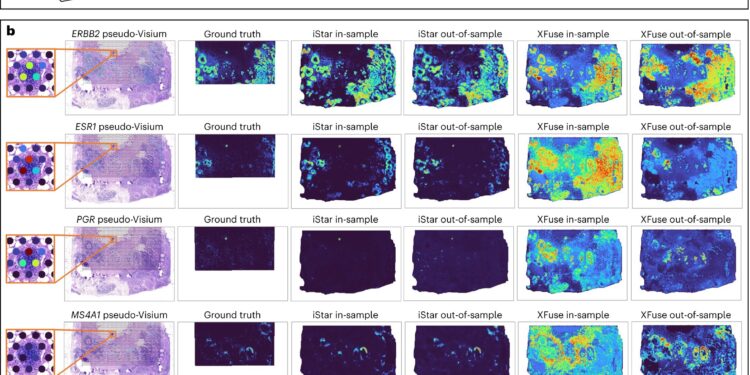
Accuracy of iStar’s super-resolution gene expression prediction and workflow. Credit: Natural biotechnology (2024). DOI: 10.1038/s41587-023-02019-9
A new artificial intelligence tool that interprets medical images with unprecedented clarity does so in a way that could allow time-strapped clinicians to devote their attention to critical aspects of disease diagnosis and interpretation. pictures.
The tool, called iStar (Inferring Super-Resolution Tissue Architecture), was developed by researchers at the Perelman School of Medicine at the University of Pennsylvania, who believe it can help clinicians diagnose and better treat cancers that would otherwise could go unnoticed.
The imaging technique provides both highly detailed views of individual cells and a broader look at the entire spectrum of human gene function, which would allow doctors and researchers to see cancer cells that otherwise might have be practically invisible. This tool can be used to determine whether safety margins have been achieved through cancer surgical interventions and automatically provide annotations for microscopic images, paving the way for molecular diagnosis of diseases at this level.
A paper on the method, led by Daiwei “David” Zhang, Ph.D., research associate, and Mingyao Li, Ph.D., professor of biostatistics and digital pathology, was published today in Natural biotechnology.
Li said iStar has the ability to automatically detect critical anti-tumor immune formations called “tertiary lymphoid structures,” the presence of which correlates with a patient’s likely survival and favorable response to immunotherapy, which is often administered for cancer and requires great precision in patient selection. . This means, Li said, that iStar could be a powerful tool for determining which patients would benefit most from immunotherapy.
The development of iStar was undertaken in the context of spatial transcriptomics, a relatively new field used to map gene activities in tissue space. Li and his colleagues adapted a machine learning tool called Hierarchical Vision Transformer and trained it on standard tissue images.
It starts by breaking down the images into different steps, starting small and looking for the finer details, then moving up and “grabbing larger tissue patterns,” according to Li. A network guided by the AI system d ‘iStar uses information from the hierarchical vision transformer to then absorb all of that information and apply it to predict gene activities, often at near single-cell resolution.
“The power of iStar comes from its advanced techniques, which mirror, conversely, the way a pathologist would study a tissue sample,” explained Li. “Just like a pathologist identifies larger regions and then zooms in on detailed cellular structures, iStar can capture overall tissue structures and also focus on the details of a tissue image.”
To test the tool’s effectiveness, Li and colleagues evaluated iStar on many different types of cancer tissue, including breast, prostate, kidney, and colorectal cancers, mixed with healthy tissue. In these tests, iStar was able to automatically detect tumor and cancer cells that are difficult to identify simply with the naked eye. Clinicians of the future may be able to detect and diagnose cancers that are harder to see or identify with iStar acting as a layer of support.
In addition to the clinical possibilities presented by the iStar technique, the tool is evolving extremely quickly compared to other similar AI tools. For example, when configured with the breast cancer dataset the team used, iStar completed its analysis in just nine minutes. In contrast, the best competing AI tool took more than 32 hours to achieve a similar analysis.
This means that iStar was 213 times faster.
“The implication is that iStar can be applied to a large number of samples, which is essential in large-scale biomedical studies,” Li said. “Its speed is also important for its current extensions in predicting d “3D samples and biobanks. In the 3D context, a block of tissue can involve hundreds or even thousands of serially cut tissue slices. The speed of iStar makes it possible to reconstruct this enormous amount of spatial data in a short time period.
And the same goes for biobanks, which store thousands or even millions of samples. This is where Li and his colleagues will next target their research and expansion of iStar. They hope to help researchers better understand the microenvironments within tissues, which could provide more data for diagnostic and treatment purposes in the future.
More information:
Daiwei Zhang et al, Inferring super-resolution tissue architecture by integrating spatial transcriptomics with histology, Natural biotechnology (2024). DOI: 10.1038/s41587-023-02019-9
Provided by the Perelman School of Medicine at the University of Pennsylvania
Quote: New AI tool helps focus faster and more precisely on precision cancer pathology and beyond (January 2, 2024) retrieved January 2, 2024 from
This document is subject to copyright. Apart from fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for information only.