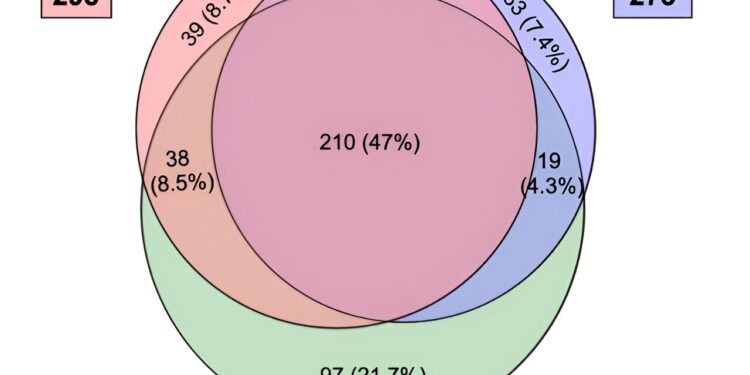
Venn diagram showing the number of miRNAs expressed in sperm samples from mice aged 3 M, 12 M, and 20 M. A total of 447 miRNAs were expressed, and approximately half of them are common. Credit: Kazusa Miyahara et al.
A recent study reported that changes in mouse sperm microRNAs caused by aging can affect the growth and development of offspring. This finding adds to the growing literature on the effects of paternal aging on offspring.
Details of the study were published in the journal Scientific reports on December 7, 2023.
Marriage and childbearing later in life are increasingly becoming the norm. While the impacts of maternal age on offspring, such as a higher risk of miscarriage and Down syndrome, are widely understood, the impacts on the paternal side are less so.
However, this is changing. Recent epidemiological studies have demonstrated that paternal aging exerts a greater influence on the increased risk of neurodevelopmental disorders such as autism spectrum disorders.
A research team led by Professor Noriko Osumi from the Department of Developmental Neuroscience at Tohoku University School of Medicine previously revealed that epigenetic factors including histone modifications in spermatogenesis and DNA methylation in mouse sperm, undergo changes with age. These modifications could lead to transgenerational effects.
However, the impact of paternal aging on microRNAs (miRNAs), small non-coding RNA molecules that play a crucial role in regulating gene expression, remains underexplored.
Correlation analyzes between ASD-associated genes whose expression levels varied across groups and their regulatory miRNAs. Two genes important for brain function, Oxtr and Gabrb2, are regulated by miR-466j (left) and miR-24-3p (right), microRNAs whose expression levels change with aging. Credit: Kazusa Miyahara et al.
To address this, the same research team conducted a comprehensive analysis of age-related variations in microRNAs in mouse sperm. They compared the microRNAs present in the sperm of mice aged 3, 12 and 20 months and identified the microRNAs whose quantity had changed.
The researchers found significant age-related differences in microRNAs. Some changes involved microRNAs responsible for regulating the nervous system and genes linked to autism spectrum disorders, and these altered microRNAs included those transferred to fertilized eggs.
“Our study reveals the potential association between alteration of sperm microRNAs caused by paternal aging, highlighting the importance of studying the impact of sperm microRNAs on offspring, an aspect that has been relatively neglected in research previous ones,” says Osumi.
It is expected that further exploration of epigenetic factors, particularly microRNAs, will not only help elucidate the pathogenic mechanisms underlying neurodevelopmental disorders, but will also provide insight into the promotion of health and disease prevention of successive generations.
Expression levels of miR-10a-5p (left) and miR-146a-5p (right), microRNAs transferred to the fertilized egg, change with age. Credit: Kazusa Miyahara et al.
Osumi points out that their study broadens the field when it comes to exploring the link between paternal age and potential child health complications. “Although age-related changes in oocytes are well documented, much of the focus has been on sperm fertility. Recognizing the myriad of epigenetic transformations associated with sperm aging, as evidenced by the microRNAs examined in this study becomes imperative.”
The findings also gain relevance in the context of Japan’s rapidly declining birth rate, necessitating integrating the perspective of sperm-related factors into the advancement of reproductive medicine.
More information:
Kazusa Miyahara et al, Investigating the impact of paternal aging on murine sperm miRNA profiles and their potential link to autism spectrum disorders, Scientific reports (2023). DOI: 10.1038/s41598-023-47878-z
Provided by Tohoku University
Quote: Mouse study finds aging sperm affects microRNAs, increasing risk of neurodevelopmental disorders (January 12, 2024) retrieved January 12, 2024 from
This document is subject to copyright. Except for fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for information only.