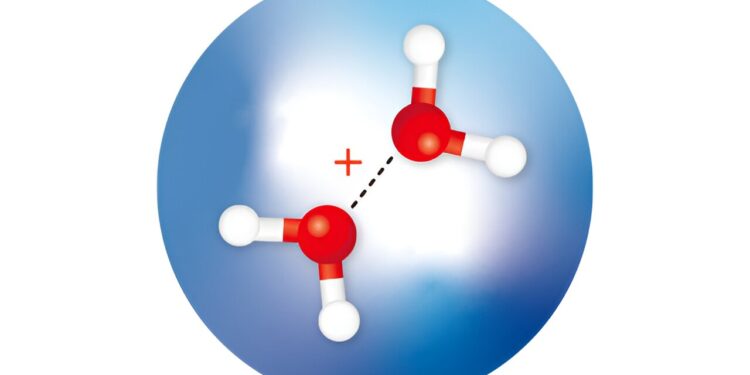
RIKEN researchers isolated and observed dimeric water cations (red spheres: oxygen atoms; white spheres: hydrogen atoms) within helium nanodroplets (large blue sphere) and determined the structures of the isomers . Credit: Adapted from The Journal of Physical Chemistry Letters (2023). DOI: 10.1021/acs.jpclett.3c02150, CC BY 4.0
An elusive structure involving two water molecules, which had been predicted but never observed, has been isolated by RIKEN chemists. This discovery could have implications in a wide range of fields from astrochemistry to metal corrosion. The article is published in The Journal of Physical Chemistry Letters.
An energetic particle or photon can remove an electron from a water molecule, creating a positive ion (cation; H2Oh+) and an electron. This ionization of water can trigger a cascade of other reactions with nearby molecules.
Water ionization plays an important role in biological processes and radiation chemistry and promotes corrosion at interfaces between water and metals. The way in which water ionization takes place is therefore a crucial question for physical chemists.
Calculations predict that after the ionization of a water molecule, two isomers of a positively charged ion of a water dimer (two water molecules loosely connected by a weak bond) will quickly form. An isomer (H3Oh+·OH) has been observed to form when a proton is transferred from one water molecule to another.
The other isomer has a half-bonded (or half-bonded) structure (H2O·OH2)+, but it has never been isolated or confirmed by spectroscopic measurements. Calculations suggest that it has a higher energy than the proton transfer dimer.
Now, Susumu Kuma of the RIKEN Atomic, Molecular, and Optical Physics Laboratory and colleagues isolated the two water dimer ions by trapping them in tiny droplets of cold helium. They also used infrared spectroscopy to determine their structures.
Kuma and his colleagues used an ultracold environment to make the isomers. The water molecules in the helium droplets cooled rapidly as the helium atoms evaporated from the surface of the droplets. This process formed the metastable hemi-bound isomer due to its very rapid stabilization within the cold droplets.
Kuma and his team then studied the coexistence of the two isomers using computational and spectroscopic methods. The spectroscopic signatures of the molecular ions were almost identical to those of bare ions, without helium surrounding them. “This finding indicates that we can directly compare measurements on bare ions with the results of quantum chemical calculations,” explains Kuma. “This greatly facilitates the structural analysis of dimers.”
This discovery will help launch further studies in this area, Kuma predicts. “The discovery of the hemi-bonded cations of water will promote further studies of the primary events that are important for understanding the radiation chemistry of water,” he says.
Kuma’s team intends to search for other structures that have not yet been observed. “We plan to increase the size of the aqueous complex cations in the helium droplets,” says Kuma. “We expect to find novel but important chemical structures in the spectra.”
More information:
Arisa Iguchi et al, Isolation and infrared spectroscopic characterization of hemilated water dimer cation in superfluid helium nanodroplets, The Journal of Physical Chemistry Letters (2023). DOI: 10.1021/acs.jpclett.3c02150
Quote: Helium droplets capture a double water structure (February 9, 2024) retrieved February 9, 2024 from
This document is subject to copyright. Apart from fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for information only.