Teplizumab delays type 1 diabetes (T1D) in at-risk people in the TN10 trial, but participants respond differently to the treatment. Systemic non-pathogenic antibody responses against intestinal bacteria can be quantified by an anti-commensal antibody test (ACAb). People with higher IgG2 responses to specific gut bacteria are more likely to get an early diagnosis of T1D and respond better to Teplizumab. Credit: Jayne Danska Tanenbaum Chair in Molecular Medicine, Senior Scientist. Deputy Chief, Faculty Development and Diversity, Hospital for Sick Children Research Institute. Professor, Department of Immunology and Department of Medical Biophysics, University of Toronto.
The microbiome offers a wealth of data on health and disease, and new findings suggest that antibodies against gut microbes may determine how well patients respond to a new monoclonal antibody drug that delays the onset of type 1 diabetes.
More and more scientists are discovering that the gut microbiome has unexpected relationships with health and disease. Research on the gut-brain axis, for example, has revealed a surprising relationship between gut microbes and mental health. But medical investigators say the list is longer and the link to gut microbes is just as complex.
Now, clinical trial data has allowed researchers to track how the gut microbiome may influence patients’ response to tepluzimab, a drug that delays type 1 diabetes. Monoclonal antibody therapy targets T cells and stops them from destroying insulin-producing beta cells. The antibody is the first treatment approved by the U.S. Food and Drug Administration to delay the metabolic disorder in high-risk individuals.
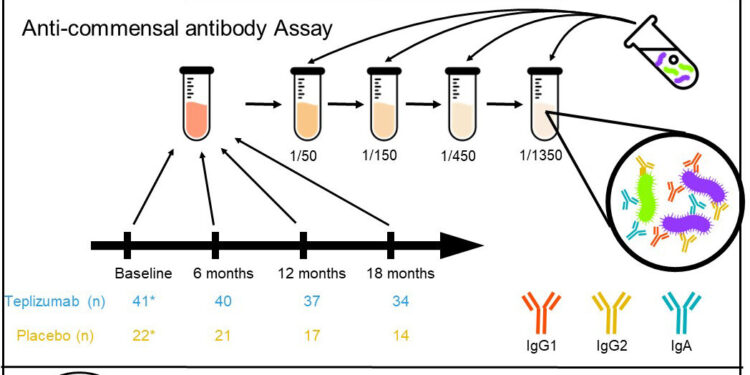
The FDA approved the drug based on the results of a randomized clinical trial known as the TrialNet-10 study, or TN-10 study for short. Medical investigators at the University of Toronto re-examined the TN-10 trial, studying more than 200 blood samples from 63 participants before and after treatment with teplizumab.
Results of the Toronto analysis, reported in the journal Scientific translational medicinesheds new light on the relationship between the immune system and the microbiome, revealing how gut microbes can shape the progression of type 1 diabetes. With this new knowledge, clinicians could better identify which patients are most likely to respond to the teplizumab.
Formerly known as juvenile diabetes because it most often appears during childhood, this condition is linked to a multitude of potential causes. The disorder is linked to a stunning immune system, which destroys insulin-producing beta cells in the pancreatic islets of Langerhans. Destruction of beta cells leads to lifelong insulin dependence.
Doctors say there are two other possible causes of type 1 diabetes: a genetic predisposition to the disease and exposure to certain viruses. Either way – faulty DNA or viral exposure – the end result is a T cell attack on the beta cells of the pancreas. Type 1 diabetes is classified as an autoimmune disease, but is more accurately defined as an autoinflammatory disease.
“Targeted immune therapies are effective for the treatment of autoinflammatory diseases,” writes Quin Yuhui Xie, lead author of a new investigation published in Scientific translational medicine. “For example, treatment with teplizumab, a T cell-specific anti-CD3 antibody, delayed disease onset in participants at high risk of type 1 diabetes in the TrialNet 10 trial.
“However, the heterogeneity of therapeutic responses in TrialNet-10 and other immunotherapy trials identifies gaps in understanding disease progression and treatment responses,” added Xie, a researcher in the Department of Biophysics medical school from the University of Toronto in Canada.
The FDA approved tepluzimab in November 2022 amid results revealing that not all patients in the TN-10 study experienced the same benefits. The reason for this discrepancy, Xie now says, can be explained by specific commensal bacteria. Commensal bacteria are called “friendly”. They constitute the microbiota, a diverse community numbering in the trillions inhabiting the mucosal and epidermal surfaces of humans. These bacteria play a critical role in defense against pathogens and apparently in response to the drug teplizumab.
“We studied anti-commensal antibody responses against a panel of taxonomically diverse gut bacteria species in the sera of TN-10 participants before and after treatment with teplizumab or placebo,” Xie wrote.
The Toronto team hypothesized that the differences in patient responses could be explained by anti-commensal antibodies directed against commensal microbes in the gut microbiome. The team then analyzed antibody profiles in 228 serum samples from 63 participants in the TN-10 trial before and after treatment with teplizumab.
Patients who had longer-lasting antibody responses against three species of gut bacteria – Bifidobacterium longum, Enterococcus faecalis and Dialister invisus – spent more time on teplizumab treatment before being diagnosed with type 1 diabetes. The data Clinical trials found that patients with a stronger immune response against all three gut microbes tended to benefit most from the drug’s disease-delaying effects.
“The gut microbiome is a potential source of biomarkers,” Xie concluded, noting that the Toronto team “previously reported that antibody responses against gut commensal bacteria were associated with the diagnosis of type 1 diabetes, suggesting that certain antimicrobial immune responses could help predict the onset of disease.
More information:
Quin Yuhui Xie et al, Immune responses to gut bacteria associated with time to diagnosis and clinical response to T cell-directed therapy for prevention of type 1 diabetes, Scientific translational medicine (2023). DOI: 10.1126/scitranslmed.adh0353
© 2023 Science X Network
Quote: Gut microbes could determine patients’ response to a drug that delays the onset of type 1 diabetes (December 27, 2023) retrieved December 27, 2023 from
This document is subject to copyright. Apart from fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for information only.