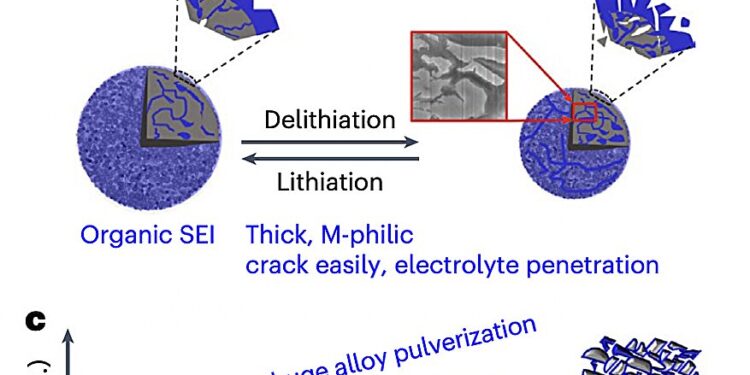
Schematic of the cycled micro-sized M-anodes LixM (M = Si, Sn, Al, and Bi) with thin inorganic LiF (magenta) and thick organic SEI (blue). a, The LiF SEI has weak binding to the LixM phases, which keeps it intact during lithiation-delithiation cycles, enabling the long cycling of micro-sized alloy anodes with restricted particle sputtering and reduced electrode swelling. b, The organic SEI has strong binding to the LixM phases and easily cracks along the contraction of the alloy particles, leading to electrolyte penetration, further SEI formation, and alloy particle sputtering with huge electrode swelling (thickness growth). c, The evolution of electrode thickness along the long cycling of alloy anodes with LiF (magenta) and organic SEI (blue). Credit: Li et al. (Natural energy(Springer, 2024).
Lithium-ion batteries (LiB) have become the most widely used rechargeable batteries in the world. Energy researchers and materials scientists have been trying to identify alternative materials that could serve as LIB components, which could potentially lead to improvements in battery performance and efficiency without significantly increasing manufacturing costs.
So far, graphite has been the most widely used anode material for LiB, due to its relatively low cost, light weight, and durability. In recent years, however, studies have identified promising alternatives to graphite-based anodes, one of which is the micrometer-sized alloy anode.
Alloy anodes are made of metal alloys that can react with lithium, such as silicon (Si), tin (Sn), or aluminum (Al). Anodes made from these alloys could have significant advantages over graphite anodes, including lower cost and the potential to increase battery capacity.
Despite their potential advantages, small-sized alloy anodes have so far proven to be less reliable than graphite anodes. This is partly because they often lead to rapid capacity degradation and low Coulomb efficiencies, particularly when combined with carbonate-based electrolytes.
Previous studies have shown that the solid electrolyte interphase (SEI), the protective layer that forms on the anode during battery cycling, bonds too strongly to the alloys. This can lead to structural cracks in both the solid electrolyte interphase and the alloy through which the electrolyte can penetrate, forming new SEI layers as the battery is charged and discharged.
The resulting rapid degradation, observed in batteries with microscopic alloy anodes, has so far limited their large-scale use and commercialization.
In an article published in Natural energyResearchers from the University of Maryland and the University of Rhode Island have presented a new asymmetric electrolyte that could improve the performance of LiB with microscopic-sized alloy anodes.
“Using nano-sized alloy anodes can improve cell cycle life, but also reduces battery life and increases manufacturing costs,” Ai-Min Li, Zeyi Wang and colleagues wrote in their paper.
“We have significantly improved the cycling performance of micro-sized Si, Al, Sn, and Bi anodes by developing asymmetric electrolytes (solvent-free ionic liquids and molecular solvent) to form LiF-rich inorganic SEIs, resulting in 90 mAh μSi||LiNi0.8Mn0.1Co0.1O2 and 70 mAh Li3.75Si||SPAN pocket cells (surface capacity of 4.5 mAh cm−2; N/P of 1.4) to achieve > 400 cycles with high capacity retention of > 85%.
Researchers have designed and synthesized a new electrolyte that could perform optimally when combined with micrometer-sized alloy anodes and high-energy cathodes. This electrolyte is based on N-methyl-N(2-methoxyethoxy)methylpyrrolidinium hexafluorophosphate, abbreviated as NMEP.
“The asymmetric electrolyte design forms LiF-rich interphases that enable high-capacity anodes and high-energy cathodes to achieve long cycle life and provide a general solution for high-energy Li-ion batteries,” Li, Wang, and colleagues wrote.
To assess the potential of their electrolyte, the team tested it in large LiB cells. Their results were very promising, as the cells achieved high capacities above 140 mAh g-1 for 200 cycles, retaining more than 85% of their capacity after 400 operating cycles.
The newly introduced asymmetric design by the researchers improves the compatibility between LiPF6 salt, a key component of LiBs, and dimethyl ether (DME) with low reduction potential, enabling the reliable formation of LiF interfaces on microscopically small alloy anodes.
In the future, it could be tested on a wider range of batteries with different anode and cathode compositions, potentially contributing to the development of next-generation battery solutions.
More information:
Ai-Min Li et al, Asymmetric electrolyte design for high-energy lithium-ion batteries with micrometer-sized alloy anodes, Natural energy (2024). DOI: 10.1038/s41560-024-01619-2.
© 2024 Science X Network
Quote:Asymmetric electrolyte design enables high-capacity anodes in lithium-ion batteries (2024, September 10) retrieved September 11, 2024 from
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. The content is provided for informational purposes only.