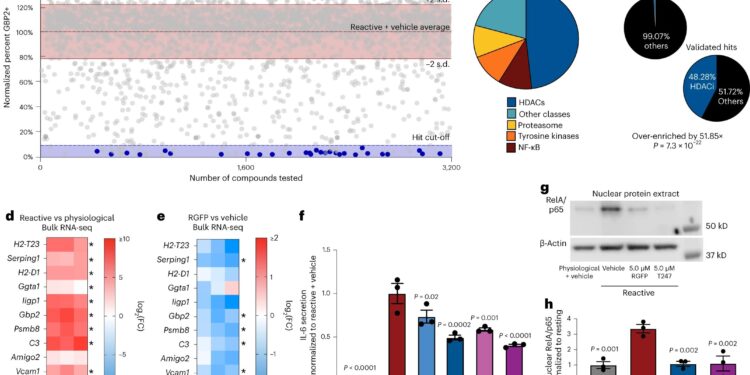
Phenotypic analysis identifies HDAC3 as a regulator of pathological reactive astrocytes. A, Scatterplot of main screen results displayed as percent GBP2 positive, normalized to reactive astrocytes and vehicle controls for all non-toxic chemicals. Validated chemicals colored blue. The blue dotted line represents the threshold for reaching a ≥90% decrease in GBP2-positive astrocytes compared to reactive astrocyte plus vehicle controls. The dashed red line represents the average percentage of GBP2 positive for reactive astrocytes plus vehicle set to 100%. Solid lines represent ±2 sd of the mean of reactive and vehicle control wells. bPie chart illustrating the chemical class distribution of the 29 validated chemical results. vsPie charts illustrating the frequency of HDAC inhibitor compounds enriched in the primary screen validated hit list compared to the primary screen chemical library as a whole, showing that HDAC inhibitors are significantly enriched in the validated hit list. P. value generated by a bilateral hypergeometric test. d,eLog heatmap2(FC) from bulk RNA-seq analysis of reagents versus physiological (d) and reactive astrocytes treated with RGFP966 (RGFP) versus vehicle (DMSO) (e). Red is upregulated and blue is downregulated. Data is presented in log form2(FC) for not = 3 biological replicates with asterisks denoting one P. < 0.05 calculated by DESeq2. F, Quantification of IL-6 ELISAs performed on media conditioned by astrocytes. Data presented as mean ± s.e.m. for not = 3 biological replicates with significance calculated relative to the reagent plus vehicle control. P.value generated by one-way ANOVA with Dunnett’s multiple comparison correction. gRepresentative Western blot image of nuclear protein extracts from physiological astrocytes, reactive astrocytes, and reactive astrocytes treated with one of the HDAC3 inhibitors RGFP966 or T247 probed for RelA/p65 and β-actin. hQuantification of experiences represented in g. Data are presented as mean ± s.e.m. for not = 3 biological repetitions (isolations of independent astrocytes). P.value generated by one-way ANOVA with Dunnett’s multiple comparison correction. Credit: Natural neuroscience(2024). DOI: 10.1038/s41593-024-01580-z
A team led by Case Western Reserve University School of Medicine scientists has identified a new therapeutic approach to combat neurodegenerative diseases, offering hope for improved treatments for Alzheimer’s disease, Parkinson’s disease, missing white matter disease and multiple sclerosis, among others.
Neurodegenerative diseases, which affect millions of people worldwide, occur when nerve cells in the brain or nervous system lose function over time and eventually die, according to the National Institutes of Health. Alzheimer’s disease and Parkinson’s disease are the most common.
The research team’s new study, published in the journal Natural neuroscience, focused on astrocytes, the most abundant cells in the brain, which normally support healthy brain function. Growing evidence indicates that astrocytes can shift to a harmful state that increases nerve cell loss in neurodegenerative diseases.
Researchers created a new cell-based technique to test thousands of possible drugs to determine their ability to prevent the formation of these unwanted astrocytes.
“By harnessing the power of high-throughput drug screening, we have identified a key protein regulator that, when inhibited, can prevent the formation of harmful astrocytes,” said Benjamin Clayton, lead author and member of the National Multiple Sclerosis Society. Society in career transition in the study. laboratory of Paul Tesar at the Case Western Reserve School of Medicine.
They found that blocking the activity of a particular protein called HDAC3 could prevent the development of dangerous astrocytes. Scientists found that by administering drugs that specifically targeted HDAC3, they could prevent the development of dangerous astrocytes and significantly increase nerve cell survival in mouse models.
“This research establishes a platform for discovering therapies to control diseased astrocytes and highlights the therapeutic potential of regulating astrocyte states to treat neurodegenerative diseases,” said Tesar, the Dr. Donald and Ruth Weber Goodman Professor of Therapeutics. innovative and principal investigator of the study.
Tesar, also director of the School of Medicine’s Glial Sciences Institute, said more research needs to be done before patients can benefit from this promising approach. But, he added, their findings could lead to the creation of new therapies that disarm harmful astrocytes and support neuroprotection, perhaps improving the lives of people with neurodegenerative diseases in the future.
“Therapies for neurodegenerative diseases typically target nerve cells directly,” said Tesar, “but here we asked whether correcting the detrimental effects of astrocytes could provide therapeutic benefit. Our results are redefining the treatment landscape for neurodegenerative diseases and open the door to a new era of drugs targeting astrocytes.
More information:
Benjamin LL Clayton et al, A phenotypic screening platform to identify chemical modulators of astrocyte reactivity, Natural neuroscience(2024). DOI: 10.1038/s41593-024-01580-z
Provided by Case Western Reserve University
Quote: Repairing unwanted brain cells could be key to preventing neurodegeneration (February 20, 2024) retrieved February 20, 2024 from
This document is subject to copyright. Apart from fair use for private study or research purposes, no part may be reproduced without written permission. The content is provided for information only.